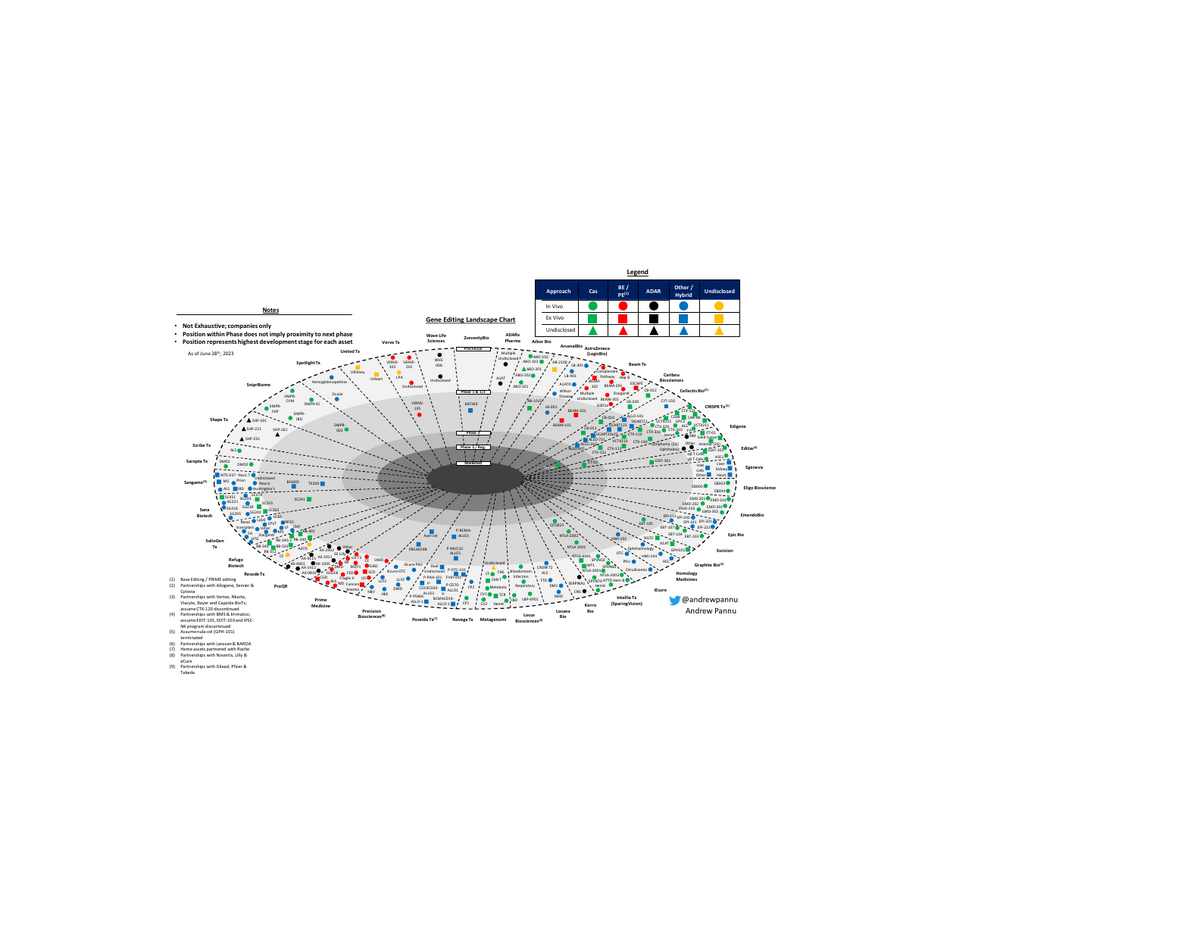
Gene Editing Landscape: 42 Companies
42 gene editing companies charted. ~85% of programs are preclinical. First CRISPR approval could be a watershed moment.
Download the Visual
I pulled together 42 public & private companies and charted the preclinical & clinical assets of each, segmented by approach & editor
Some takeaways:
- In the context of therapeutics, gene editing is the insertion, deletion or replacement of sections of a patient's DNA to treat disease. This differs from gene therapy, which is the correction, removal or addition with exogenous genetic material that does not integrate into the patient's genome
- While the below analysis is not exhaustive (there are >250 active gene editing programs), it does help illustrate how early the space is. ~85% of all programs are preclinical, with everything else in Ph1/2 except $CRSP & $VRTX exa-cel (submitted BLA, PDUFAs in Dec'23 & Mar'24)
- Things to watch as the space matures:
(1) Patient & indication selection: 1st gen assets primarily targeted severe patient populations of monogenic diseases (i.e. SCD, TDT) with limited treatment options
Emerging assets are targeting more competitive spaces (CV, Onc, Metabolic). It will be interesting to track patient / physician acceptance of a potentially curative but relatively new treatment vs. more established chronic treatments
(2) Reimbursement: as with gene therapies, the one-time nature of treatment will command sky-high price tags
Expect payor pushback in more prevalent conditions with an established, chronic & cheaper SoC. Companies should get comfortable with value-based agreements, where reimbursement is tied to treatment milestones & staged over time. Emerging mktd. gene therapies will help set guidelines here (i.e. Roctavian)
(3) CMC: this can quickly become a bottleneck for companies during clinical development & beyond, as scaling these novel processes to pivotal / commercial grade with appropriate comparability to earlier-stage trials is difficult
(4) Safety: given we are talking about permanent genome edits, this will remain in focus for the foreseeable future
The FDA has been cautious and placed lengthy clinical holds on several programs to request more data / investigate abnormalities. Management of off-target effect & immunogenicity is key
- Since CRISPR-Cas9 was invented in 2012, gene editing companies have raised >$18B (public & private). Approval of $CRSP / $VRTX exa-cel in 2023 would thus be a watershed moment for the space, and the culmination of a decade of fast-paced innovation & investor / pharma enthusiasm
- Looking ahead beyond exa-cel, there are a few other catalysts to monitor:
- Additional NTLA-2001 data in ATTR & initiation of global pivotal study ($NTLA)
- First look at clinical data for a base editor via VERVE-101 ($VERV)
- Clinical update from EDIT-301 TDT trial ($EDIT)
- To see my prior breakdown of the gene therapy landscape, check out: https://rb.gy/6h7qh

