IL-6 Cardiovascular Landscape: 101 Programs, 3 Control the Window
101 IL-6 programs mapped. Only 4 target cardiovascular indications. Three late-stage players control the 2027-2029 approval window.
Download the Report
The IL-6 pipeline contains 101 named programs, but only 4 target cardiovascular indications. Of 55 preclinical programs, just 1 pursues CV. The rest focus on rheumatoid arthritis, oncology, and autoimmune diseases.
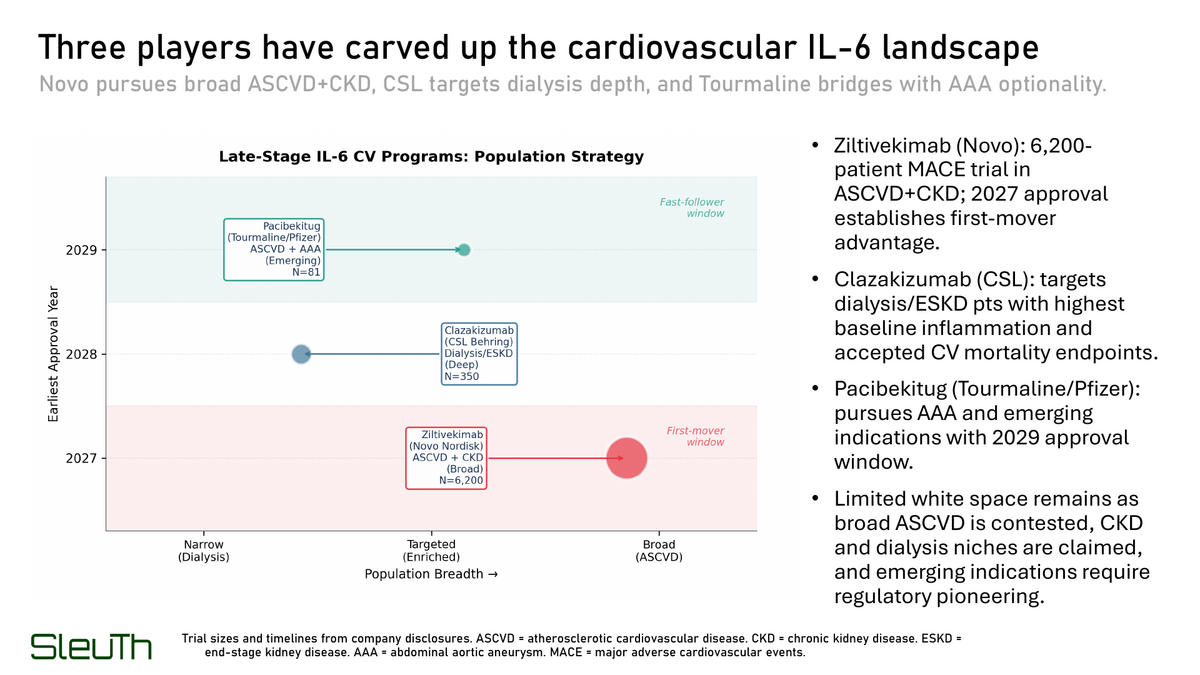
Three programs control the 2027-2029 CV approval window. Novo Nordisk's ziltivekimab is running a 6,200-patient MACE trial in ASCVD+CKD with a 2027 approval timeline. CSL's clazakizumab targets dialysis/ESKD patients with the highest baseline inflammation. Tourmaline/Pfizer's pacibekitug pursues ASCVD and AAA with a 2029 window. Preclinical entrants cannot catch this timeline.
All IL-6 programs achieve 85-94% hs-CRP reductions regardless of indication, eliminating biomarker superiority as a differentiation lever. Approved IL-6R antagonists hit 92-94%. CV-focused programs (ziltivekimab 88%, pacibekitug 86%, clazakizumab 90%) fall within the same band. Competition must occur on population selection or endpoint innovation.
HFpEF and abdominal aortic aneurysm have zero IL-6 clinical programs despite biological rationale. These emerging indications offer genuine differentiation but require sponsors to pioneer regulatory endpoints without precedent.
Preclinical programs should avoid broad ASCVD pursuit and instead target high-inflammation subgroups (CKD/dialysis enrichment) or emerging indications where late-stage competitors have not yet established dominance.

