The ALS Development Field Is Trapped at Two Extremes
Shionogi just spent big consolidating edaravone rights. But here's the strategic tension hiding in plain sight.
Download the Visual
Shionogi just spent big consolidating edaravone rights - validating that pan-ALS treatment is commercially viable. But here's the strategic tension hiding in plain sight.
I used Sleuth to map 623 ALS programs and found the field splitting into two opposing camps, both with fatal flaws.
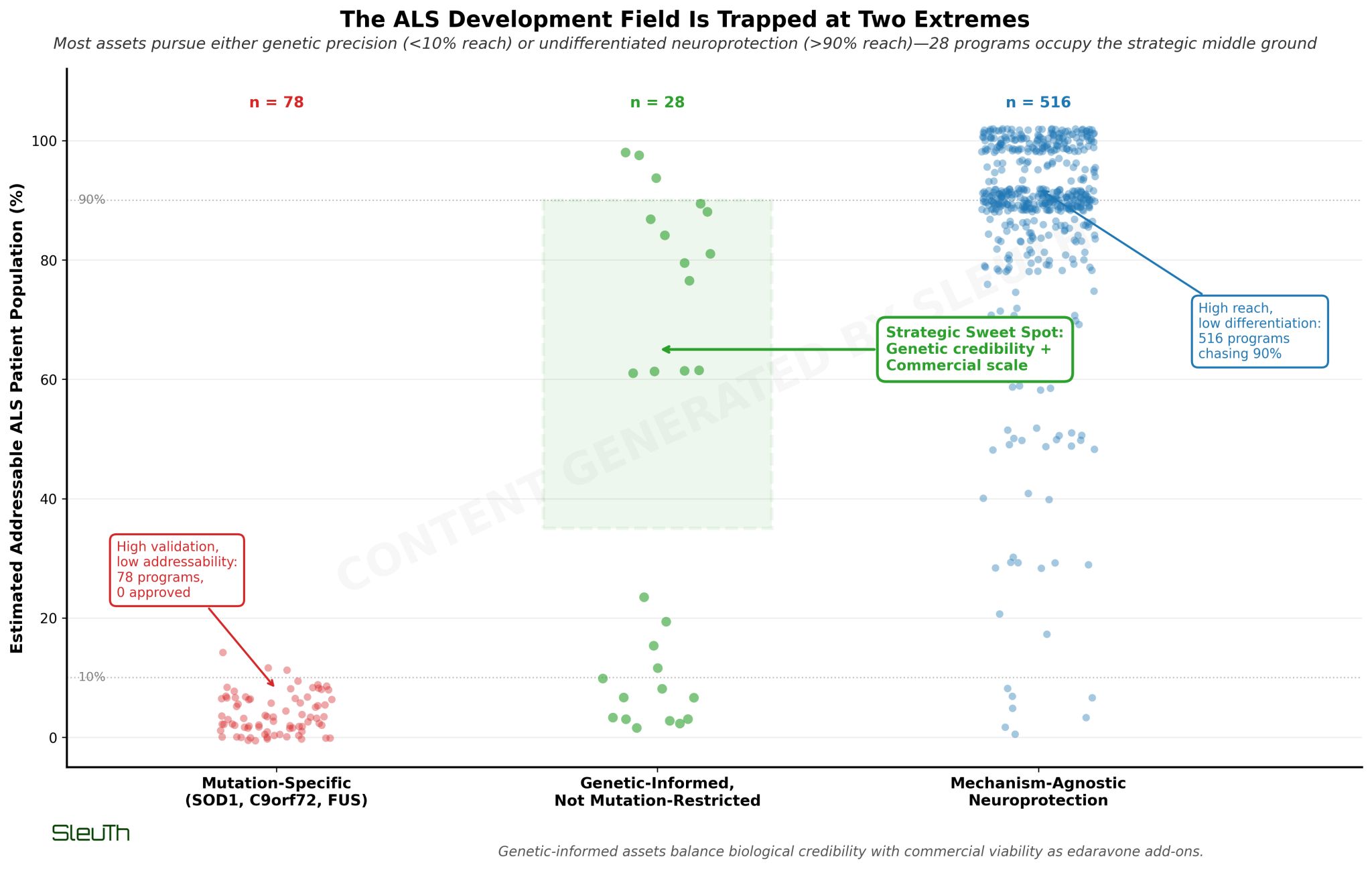
Camp 1: Gene therapies targeting specific mutations (SOD1, C9orf72, FUS). These cover only 10% of ALS patients. The field has 67 gene therapy programs - 72 are preclinical, zero approved. High scientific confidence, but even with perfect execution you're capped at single-digit market share.
Camp 2: Broad neuroprotection approaches (antioxidants, anti-inflammatories). These target 90% of patients. 192 small molecule programs are here, but most can't explain why they should work better than existing options. You get market size but lose differentiation.
The data reveals a third category being overlooked: 28 programs that are genetically informed but not mutation-restricted. Examples: drugs targeting TDP-43 protein clumps or repeat RNA toxicity. These mechanisms were discovered in familial ALS but turn out to be broken in most sporadic cases too.
This is the strategic sweet spot. You maintain biological credibility (it's not just "try another antioxidant") while addressing the full patient population. And you're positioned as complementary to edaravone, not competitive.
The portfolio implication: BD teams should stop forcing a choice between genetic precision and broad reach. The winning assets will be those with genetic validation that scale beyond rare mutations.

