A $50B CHIPS-Like Act for Domestic Drug Manufacturing?
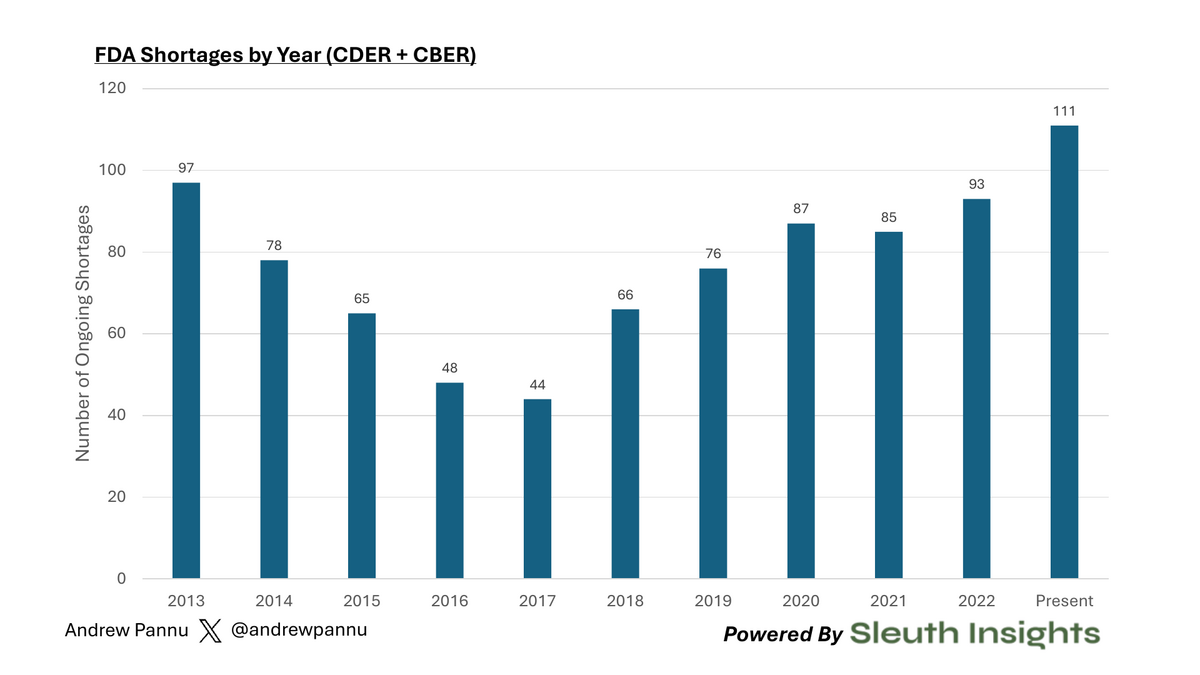
The number of active drug shortages in the US is at a 10+ year high.
Download the Visual
The number of active drug shortages in the US is at a 10+ year high:
Fig 1: FDA Shortages by Year
Why? Mainly the economics of generics (note: biosimilars are different):
- Manufacturers face severe margin compression, with >80% price cuts as multiple players flood the market
- In exchange for preferred formulary tiering, PBMs will often extract additional cost savings, meaning the way to win is through volume
- Given the cost to manufacture in the US is 2-3x higher than overseas markets such as India, sometimes the price falls too low to make a profit
- Over time, only a few manufacturers may be left, leaving that market exposed should one exit or demand spikes. It may take months for other manufacturers to pivot their facilities and bridge the gap - thus leading to shortages in the interim
A study by IQVIA shows that economics drive most shortages, as nearly all of them occur when the avg. invoice price is $5 or less:
Fig 2: Average Price
And when there is supplier concentration in the market:
Fig 3: Market Concentration
But despite the poor economic incentives, generics are vitally important - they make up >90% of all prescription drugs scripts. They save the US healthcare system >$380B annually and they build our armamentarium of low-cost medicines for future generations
Over the past ~10 years, the # of FDA-registered generic API manufacturing facilities fell by (18%) in the US and (13%) in the EU; over the same period, the number increased +21% in India and +2% in China.
Foreign generic manufacturers are less under the FDA's oversight and poor QA could lead to deadly drug impurities or imbalances vs. the brand which impact patients' health.
Fig3: 67% of FDA Form 483s in 2024 are for India-based Companies
And for the US, a big concern is the types of drug classes exposed to foreign market concentration: antibiotics, chemotherapies, etc. ~90% of generic sterile injectables used for critical acute care in the US rely on starting materials and APIs from China & India.
Which brings us back to the CHIPS Act:
The CHIPS Act authorized ~$53B for on-shoring semiconductor manufacturing to improve supply chain resilience and national security
There are similar concerns around API manufacturing, and those dollars could go far - whereas semiconductor facilities cost billions, some estimates peg a sterile injectables plant at $300M
The money could also be used to shore up broken incentives:
- Long-term procurement contracts for on-shoring manufacturing, particularly for critical drugs
- Better reimbursement terms under Medicare
- Payments to hold extra inventory
- Credits for adoption of continuous manufacturing principles (vs. batch manufacturing)
The BIOSECURE Act is the first sign that legislation is coming. It will take a few years, but I wouldn't be surprised if the final bill contains similar investment incentives for domestic drug manufacturing as we saw with the CHIPS Act

